Selective modulation of a key receptor found on mast cells and sensory neurons.
MRGPRX2 Antagonist
Blocking MRGPRX2 has the potential to be the first oral treatment for a variety of mast cell-mediated diseases, including chronic spontaneous urticaria. In addition, due to its unique function on peripheral sensory neurons, blocking MRGPRX2 could provide fast relief of itch associated with inflammatory diseases, such as atopic dermatitis.
Our CIndU Phase 2 trial was completed in May 2025 and generated data demonstrating clinical activity in a patient population with symptomatic dermographism. Given significant overlap between the diseases and patient populations, along with the contribution of neurogenic inflammation, we believe these data strongly support the role of MRGPRX2 in CSU and AD program.
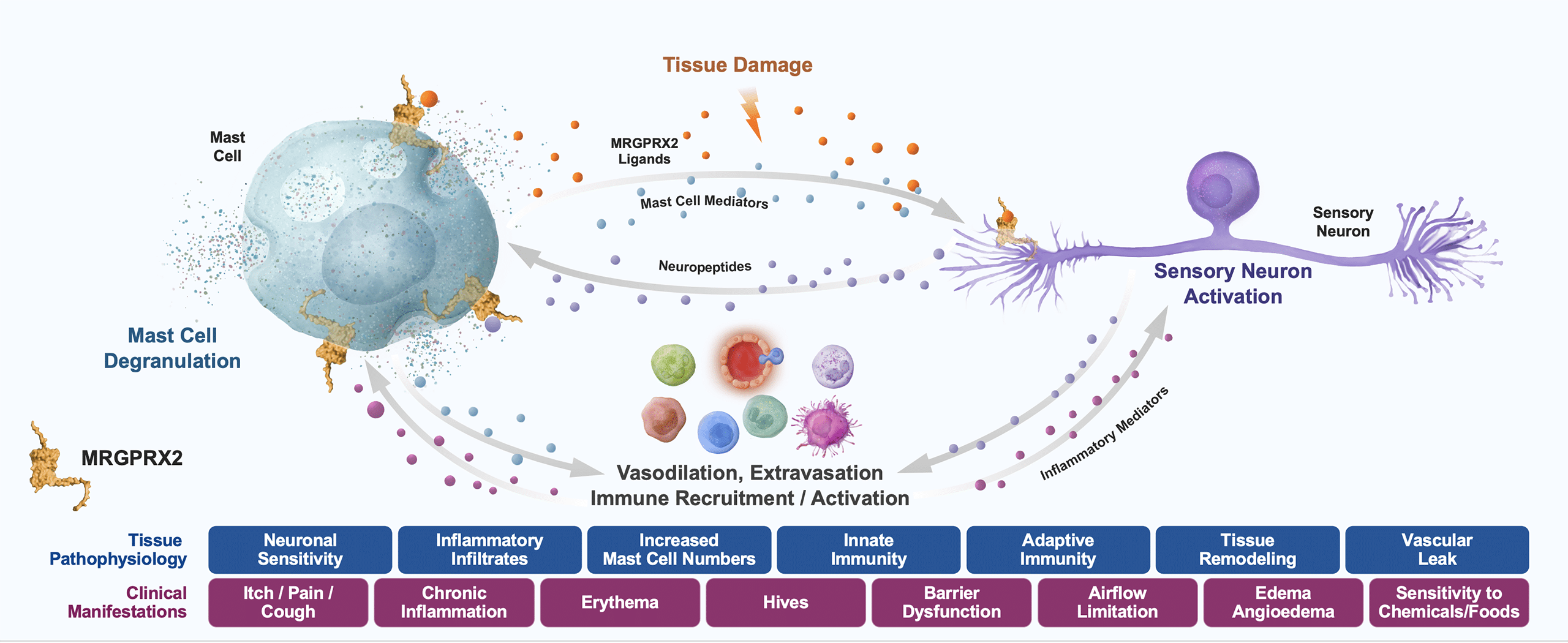
- Chronic Spontaneous Urticaria: Driven by underlying mast cell activity, these patients will develop spontaneous and recurrent hives and angioedema without a known environmental trigger, occurring on any part of their body. Current treatments offer only symptomatic relief. Blockade of the MRGPRX2 receptor and its subsequent downstream effects provide a novel target for the treatment of this pathology.
- Atopic Dermatitis (AD): Mast cells with MRGPRX2 positivity have been implicated in atopic dermatitis and are known to be in close proximity to peripheral sensory neurons, which also play a role in the itch associated with AD. AD has both an inflammatory component as well as upregulation of neurons creating itch, therefore targeting MRGPRX2 may play a significant role in the treatment of AD by targeting both aspects.
- Migraine: Migraine is a neurological disorder, estimated to affect more than 40 million people in the United States and more than 10% of the global population. More than 10 million Americans are eligible for preventive therapy, yet current treatments are limited to only improving migraine days per month by approximately two days more than placebo. MRGPRX2 is expressed by both human trigeminal neurons and meningeal mast cells, positioning it as a potential key mediator of neurogenic inflammation in migraine. Activation of trigeminal afferents and meningeal mast cells is central to migraine initiation and propagation, and in vivo preclinical headache models support a pathogenic role for MRGPRX2 signaling in driving migraine-like pain behaviors. Given that MRGPRX2 can be activated by multiple endogenous and exogenous ligands, antagonism of this receptor represents a broader upstream approach to dampen aberrant trigeminal activation and mast cell–mediated neuroinflammation. We have shown that EVO756 blocks a range of relevant MRGPRX2 ligands, including PACAP, supporting its potential to reduce migraine frequency across a broad patient population, including individuals who do not achieve sufficient benefit with existing preventive therapies.
- Other Indications: We believe EVO756 has the potential to address several additional chronic inflammatory diseases. Based on our initial research and preclinical data, we are exploring the potential for EVO756 in asthma, interstitial cystitis, irritable bowel syndrome and pruritus (itch) as potential expansion indications due to their large unmet patient needs along with the relevance of mast cells and sensory neurons in these diseases’ pathology.